Table of ContentsClose
Hospital staff around Iowa had been bracing for the day COVID-19 would hit home. Given its rapid spread across the U.S., its arrival in the state was only a matter of time.
For University of Iowa Hospitals & Clinics, that day came on March 10, when hospital leaders got word that the state’s first patient diagnosed with COVID-19 would be arriving in the Emergency Department (ED).
“What I explicitly remember about that night is watching our staff get gowned up and going in to see the patient. No one seemed like they were anxious or nervous,” says Chief Medical Officer Theresa Brennan, MD (95R, 01F, 02F). “They did what they normally do, which is take great care of the patient.”
5 lessons from UI Health Care faculty and staff
Following are the top five lessons UI Health Care faculty and staff have learned during their response to the COVID-19 pandemic.
Be prepared early and quickly

As the first patient was admitted, clinicians sent moment-by-moment radio updates to senior leaders who were onsite in the command center for the Hospital Incident Command Systems (HICS).
HICS, a management structure consisting of leaders from across the institution, had been activated following weeks of coronavirus surveillance by hospital epidemiologists and other members of the Bio-Emergency Response Team. HICS leadership enacted new procedures in response to quickly changing circumstances related to COVID-19.
Safety and Security staff cleared the hallways, making sure no patients, visitors, or staff were on the route to the Special Pathogens Unit, a secluded, negative-pressure room designated for patients with infectious diseases. This transfer was mapped out weeks in advance and involved many team members, including doctors, nurses, and Environmental Services staff.
"The team had done such a great job in preparing that it gave the people that were in the trenches, actually taking care of the patient, some peace of mind," Brennan says.
Since the arrival of the first patient with COVID-19, UI Hospitals & Clinics has cared for more than 500 people in its inpatient units—about 40% transferred from other hospitals—and screened roughly 40,000 others via telehealth.
Senior leaders at UI Health Care have shifted to all matters related to COVID-19, whether it’s policymaking, establishing best practices, implementing necessary changes, or coordinating communications.
What’s been even tougher, Brennan says, have been the decisions to restrict hospital access—unprecedented but necessary measures to protect patients, visitors, and staff.
“We’ve made a lot of changes to people’s lives, including how they enter the hospital. Despite these burdens, people have been understanding. That’s not surprising—Iowans are great,” she says.
Treat patients early, monitor often
About 80% of people who are diagnosed with COVID-19 do not need to be hospitalized.
But for those who need a higher level of care, getting hospitalized sooner may benefit them in the long run, according to Brennan.
Early detection is the first step.
To test patients rapidly and safely, the Respiratory Clinic was conceived, launched, and planned within a week.
Telemedicine allows providers to screen those with COVID-19 symptoms and determine if testing is necessary while also minimizing traffic into the hospital and limiting the spread of the virus. And once the UI Department of Pathology created its own in-house tests, UI Hospitals & Clinics was able provide same-day, drive-thru testing.
“Rapid and accurate testing is one of the keys to mitigating the spread of COVID-19 in the community,” says Katie Imborek, MD, director of offsite primary care. “I cannot overstate the importance of early identification of COVID-19 positive patients, followed up by comprehensive contact tracing and patient education.”
If patients are concerned about worsening shortness of breath, fatigue, or dehydration, they can also visit the Respiratory Clinic for an in-person evaluation with a provider. As of mid-September, over 50,000 patients have received care in the clinic.
For patients who don’t need inpatient care, the Home Treatment Team was set up to monitor them daily as they self-quarantine at home.
“It’s like the doctor is rounding on you while you are in the comfort of your own home,” says Bradley Manning, MD, clinical assistant professor of internal medicine and a hospitalist with UI Health Care.
Patients are mailed a monitoring kit, containing a blood pressure cuff and a pulse oximeter, along with instructions for self-isolation and which symptoms to monitor. They also receive daily phone calls from a nurse.
For a majority of patients, this approach proved to be crucial in helping them beat the disease while at home rather than having to come into the hospital. As of mid-August, the HTT team had cared for approximately 625 patients, a majority of whom were able to recover at home. That, in turn, reduced the risk of transmission and kept more hospital beds available for more severe cases.
Providers were also able to track those who needed more intensive levels of care and get them to the hospital sooner.
Covid by the Numbers
Patients received care in Respiratory clinic
COVID-19 adult inpatients
Video Visits (COVID-19 screening)
Statistics as of 9/21/20
Create opportunities for cross-training
During the first few weeks of the pandemic, patient volumes fell as elective and non-emergent procedures were postponed. At the same time, ICU beds remained full, raising concerns about adequate staffing levels.
To prepare for a potential surge in hospitalized patients with COVID-19, UI Health Care nursing leadership trained nearly 1,420 staff nurses to provide more intensive levels of care.
“Our staff is expanding their skill set to go where the patient need is,” says Lou Ann Montgomery, PhD, MAN, RN, NPD-BC, RN-BC, director of nursing professional development and advanced practice.
For example, intermediate-level nurses were ‘up-trained’ to provide care for high-acuity and ventilated patients in ICUs. Some outpatient nurses were trained to answer calls from community members with COVID-19-related concerns. Staff completed online courses, attended simulation labs, and trained on the unit they would be reassigned.
“It just offers us a lot of flexibility,” Montgomery says. “We have to be nimble, and we have to cross-train people to be able to work in different areas. I think we’re going to see more cross-training as we go forward.”
Use evidence-based care, don’t dismiss what you already know
Learning from peers in other states who saw early surges, the Medical Intensive Care Unit (MICU) team has always been firm in its reliance on evidence-based critical care for COVID-19 patients.
“While this disease clearly has some differences, the similarities far outweigh the differences in what we’re used to treating,” says Kevin Doerschug (94MD, 97R, 98F, 02F, 02MS), medical director of the MICU.
While many people with COVID-19 have mild or no symptoms, the disease can cause fluid buildup in the lungs and decreased oxygen flow into the bloodstream.
In the MICU, advanced oxygen-delivery devices are typically used first to try to prevent a patient from ending up on a ventilator. Really good supportive care—which often includes prone ventilation, respiratory therapy, and physical therapy—also are important. If a ventilator is needed, the team recognizes the importance of good ventilator management to optimize settings for the patient and prevent further injury to the lungs.
ECMO, or extracorporeal membrane oxygenation, may also be an option for some of the sickest patients who need to let their lungs rest and heal. UI Hospitals & Clinics, with decades of experience in providing ECMO support for the management of life-threatening cardiopulmonary failure, is one of 31 ECMO Centers of Excellence in the world with gold-level standing.
“It’s not uncommon for us to have patients on a ventilator or ECMO support be able to walk down the hallway,” says Doerschug, highlighting the value of using physical therapy specialists to keep patients mobile during treatment.
“Our interdisciplinary collaboration has always been good, but it’s been brought to a new level,” says Austin Kannegieter, BSN, RN, MBA, nurse manager in the MICU. “We really have done very well in caring for our patients. That’s been very encouraging, but we wouldn’t be able to do that without collaboration, communication, and streamlined practices.”

Pregnant with COVID-19
ECMO experts, OB-GYN specialists collaborate on life-or-death case
Monitor and treat long-term effects of COVID-19
With over 60,000 Iowans testing positive for COVID-19 since the pandemic began, there is a growing need for specialized follow-up care for a subset of patients experiencing long-term effects from the disease. In June, UI Health Care became the first health system in the state, and one of the first in the country, to establish a dedicated post-COVID-19 clinic.
The Respiratory Illness Follow-Up Clinic serves as a statewide resource to proactively identify and treat those with lingering symptoms from COVID-19.
Already evidence is showing that COVID-19 may cause pulmonary scarring, myocardial damage, decreased ejection fractions of the heart, and sometimes large and small arterial and venous occlusions. The result is ongoing symptoms that may manifest as a persistent dry cough, chronic fatigue, or decreased cardiopulmonary fitness.
“We have a strong suspicion that these long-term symptoms, which patients may experience four to six weeks after diagnosis, might be indicative of a more serious inflammatory airway disease that needs to be aggressively treated,” says Alejandro Comellas, MD, medical director of the clinic.
Comellas, who is a pulmonologist and critical care specialist, plans to establish a pulmonary registry of voluntary post-COVID-19 patients to better identify and track long-term effects of the disease and evaluate promising therapeutics.
“Our goal with the establishment of this clinic is to truly raise awareness among physicians and the community that there can be serious long-term effects of COVID-19 that must be addressed,” he says.

On the frontlines of COVID-19 research
UI mouse model tool proves crucial to COVID-19 research worldwide
When the COVID-19 pandemic took hold and researchers around the world scrambled to understand the new virus, they ran into a serious problem. Lab mice, the standard experimental animal for studying disease and testing treatments, don’t get COVID-19.
Fortunately, researchers Stanley Perlman, MD, PhD, and Paul McCray (81MD, 84R, 88F) had an answer. More than a decade ago, the pair had created a transgenic mouse with humanized ACE2 receptors that was susceptible to SARS, a close cousin of the new disease, in order to study potential coronavirus treatments.
The Jackson Laboratory, the world’s largest supplier of research mice, reintroduced and bred this UI-developed mouse model that scientists around the globe can use.
The UI team has sprung into action again, aiming to create new mouse models that will help researchers better understand COVID-19 and the immune system’s response.
They have developed a gene therapy approach that can convert any lab mouse into one that can be infected with SARS-CoV-2 and develop COVID-like lung disease. This off-the-shelf model allows labs to create their own COVID-19 mouse model within a matter of days.
Reporting in Cell, the team showed that mice treated with this gene therapy could be used to evaluate potential vaccines and therapies, including a preventive strategy known as poly I:C and the anti-viral drug remdesivir. In each case, the therapies prevented weight loss, reduced lung disease, and increased the speed of virus clearance in the mice. The mice can also be used to study important immune responses involved in clearing the SARS-CoV-2 virus.
UI Health Care site of COVID-19 international vaccine clinical trial
In July, the University of Iowa joined a large-scale, international clinical trial to study the safety, tolerability, immunogenicity, and efficacy of vaccines against COVID-19.
The study is testing an experimental RNA vaccine developed by drug companies BioNTech and Pfizer. It aims to enroll approximately 44,000 adults, with the UI site enrolling 250 participants.
“Based on our understanding of vaccines and what we have seen in some of the early-phase studies, my best guess is that we’re going to have a vaccine that works and is safe,” says Patricia Winokur, MD (88R, 91F), who is the principal investigator for the UI trial. “What we can’t say yet is whether it will protect 50% of people, 70% of people, or 90% of people from getting infected.”
It is also unclear how long the vaccine will provide immunity, which is why extensive testing is necessary even after a vaccine is approved.
“In our study, we will keep following and documenting antibody titers, safety, and the ability to prevent COVID-19 infections for two years, which is really the icing on the cake,” says Winokur, an infectious disease specialist and professor of internal medicine. “Even if a vaccine is approved, this is kind of a long-haul situation where we're really going to be looking to learn more about the vaccine and the body’s immune response as well as learning about the virus itself.
Winokur, who is also executive dean of the UI Carver College of Medicine, notes that it was crucial to enroll a diverse population to ensure that the vaccine is equally effective in people of different ages, races, and genders.
Over six visits, the participants will be evaluated to learn about the safety of the vaccines as well as any side effects that might occur. Individuals will also be surveyed weekly to see if they have developed symptoms of COVID-19.
UI to participate in clinical trial of COVID-19 vaccine
University of Iowa is participating in a large-scale, international clinical trial to study the safety, tolerability, immunogenicity, and efficacy of vaccines against COVID-19
Testing new treatment approaches: Remdesivir and convalescent plasma
As medical professionals worldwide urgently explore treatment options for COVID-19, University of Iowa Health Care has joined the quest with studies to test the effectiveness of remdesivir, an intravenous antiviral drug, and convalescent plasma transfusions.
Infectious diseases specialist Dilek Ince, MD (06R, 08F, 09F), and her colleagues saw potential in remdesivir, which had previous success in laboratory tests against the coronaviruses that cause SARS and MERS.
Following positive results from an NIH-led study in late April, the U.S. Food and Drug Administration issued emergency use authorization to expedite the drug’s availability to more patients with severe cases of COVID-19.
Under Ince’s leadership, UI Hospitals & Clinics was one of approximately 180 sites worldwide that participated in a pair of clinical trials sponsored by Gilead Sciences to test remdesivir in patients with moderate and severe cases of COVID-19. In June, Gilead Sciences announced topline results from the phase 3 clinical trial in patients with moderate COVID-19. The study found that patients in a five-day remdesivir treatment group were 65% more likely to have clinical improvement at day 11, compared with patients receiving standard of care.
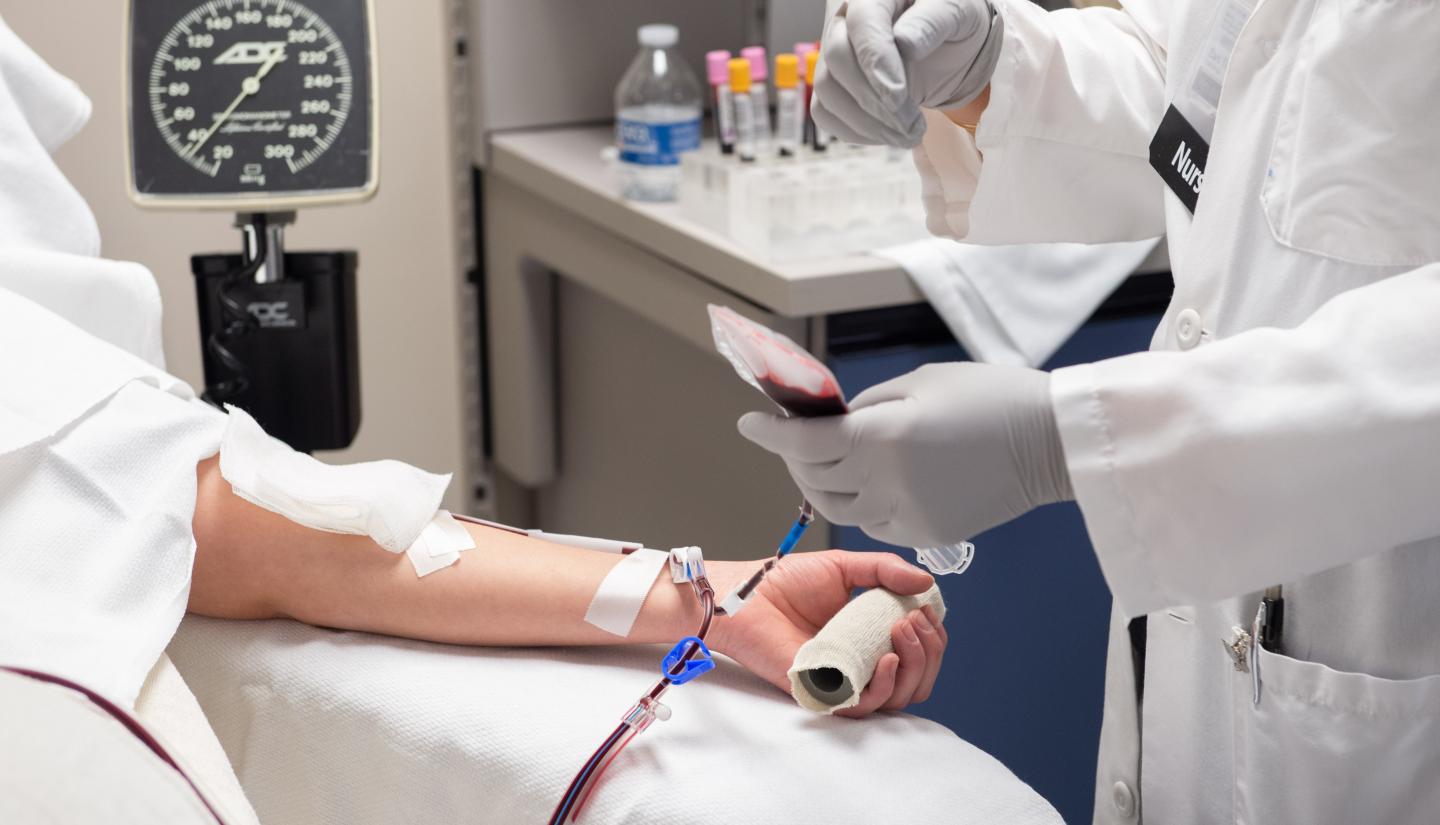
On another treatment front, UI Health Care researchers, led by Patricia Winokur, MD (88R, 91F), professor of internal medicine, and Michael Knudson, MD, PhD, clinical professor of pathology and radiation oncology, have enrolled over 100 recovered COVID-19 patients in a clinical trial to determine if plasma from these donors can help hospitalized patients with COVID-19 recover from the disease.
The key to this “convalescent plasma” approach are the antibodies produced in people when they fight off COVID-19. These antibodies can be collected in plasma donations from the recovered patients and transfused into hospitalized patients with the goal of helping those patients fight the disease more effectively.
The FDA issued an emergency use authorization for the treatment in late August after preliminary data showed the treatment is safe and may improve COVID-19 treatment outcomes, although the agency has asked for additional data to support the efficacy.
Nearly 100 patients have received plasma transfusions as part of their care for COVID-19 at UI Hospitals & Clinics. The treatment appears to be well tolerated in patients and investigators are continuing to examine outcome data to determine if the treatment has been effective, according to Knudson.
